Shanghai's New AI Model Speeds Up Antiviral Drug Breakthroughs
Shanghai's AI Breakthrough in Antiviral Research
Shanghai researchers have developed an artificial intelligence tool that could slash the time needed to create new antiviral medications. The ViraHInter model, created by a collaboration between Shanghai Artificial Intelligence Lab and several leading medical institutions, offers a digital shortcut in the race against evolving viruses.
How ViraHInter Changes the Game
Traditional drug development requires painstaking laboratory work to understand how viruses interact with human proteins. ViraHInter eliminates much of this trial-and-error by making accurate predictions about these interactions before researchers ever step into a lab.
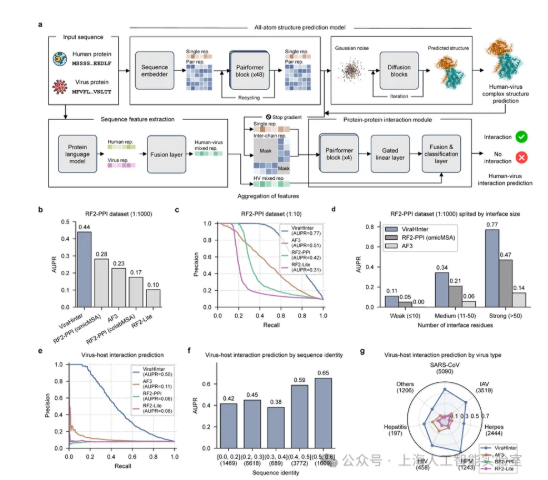
"What sets this model apart is its dual focus," explains Dr. Li Wei, a computational biologist involved in the project. "While most systems analyze either protein sequences or structures, ViraHInter does both simultaneously - like reading a book while also understanding how the pages are bound together."
The AI achieves this through two key approaches:
- Generating detailed 3D models of virus-protein interactions at the atomic level
- Using language processing techniques to spot patterns that persist even as viruses mutate
Real-World Results
Initial tests show remarkable promise. When predicting virus-human protein interactions, ViraHInter achieved an accuracy score of 0.50 - 4.5 times better than AlphaFold3, currently one of the most advanced systems in this field.
Researchers put the model to work analyzing three influenza subtypes. It successfully identified 33 host factors shared across all three strains - potential weak spots that could lead to broader-spectrum treatments.
Perhaps most exciting is the model's ability to tackle new threats. Even when presented with completely novel virus sequences, ViraHInter maintained strong performance. This adaptability could prove crucial when the next pandemic emerges.
What This Means for Future Treatments
The implications extend beyond influenza. The same principles could apply to coronaviruses, HIV, and other rapidly-mutating viruses that have historically challenged drug developers.
"We're not just speeding up the process," notes Dr. Chen from Ruijin Hospital. "We're uncovering targets that traditional methods might miss entirely. Some of these interaction points are so subtle they'd be nearly impossible to spot without this level of computational power."
As antiviral resistance grows worldwide, tools like ViraHInter could help scientists stay one step ahead of evolving pathogens. The research team is now working to integrate their findings into actual drug development pipelines.
Key Points
- Dual analysis: Combines protein structure and sequence data for more complete predictions
- Speed advantage: Reduces reliance on time-consuming lab experiments
- Broad applications: Effective against multiple virus types, including emerging strains
- Proven performance: Outperforms existing models by significant margins
- Drug development: Identifies promising treatment targets that might otherwise go unnoticed