AI Breakthrough Speeds Up Antiviral Drug Discovery
New AI Model Predicts Virus Behavior with Unprecedented Accuracy
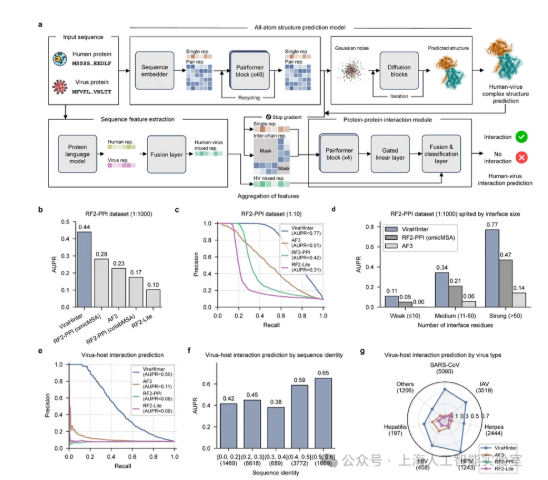
Scientists in Shanghai have developed an artificial intelligence system that could dramatically accelerate the creation of antiviral drugs. The ViraHInter model, a collaboration between Shanghai AI Lab and several leading medical institutions, eliminates the need for time-consuming lab experiments by predicting how viruses "hijack" human proteins digitally.
How ViraHInter Works Differently
Traditional methods analyze either protein sequences or 3D structures separately, but ViraHInter's breakthrough comes from examining both simultaneously. Imagine trying to understand a lock-and-key mechanism by either just looking at the shapes or just reading the parts list - this AI does both at once.
The system generates detailed atomic-level models of virus-human protein interactions while also identifying patterns that remain consistent even as viruses mutate. This dual approach means researchers can:
- Design drugs that target stable viral features less likely to change
- Visualize exactly how potential treatments would interact with viruses
- Respond faster to emerging viral threats
Performance That Turns Heads
Benchmark tests revealed staggering results. ViraHInter achieved a prediction accuracy of 0.50 for virus-human protein interactions - 4.5 times better than AlphaFold3, the previous gold standard. When analyzing three influenza subtypes, the model pinpointed 33 shared host factors that could serve as universal treatment targets.
"What's truly exciting," explains a researcher familiar with the project, "is how well it handles novel viruses. Even with limited sequence data, ViraHInter maintains high accuracy - crucial for responding to the next pandemic."
Implications for Future Treatments
The technology shines brightest where we need it most: combating rapidly evolving viruses. By identifying conserved viral patterns and predicting interactions for unknown pathogens, ViraHInter could:
- Shorten development timelines for flu and coronavirus medications
- Reveal treatment targets that work across multiple virus strains
- Provide crucial early data when new diseases emerge
As one scientist put it: "This isn't just an incremental improvement - it's changing how we approach antiviral research altogether."
Key Points
- Dual analysis: Examines both protein sequences and 3D structures concurrently
- 4.5x improvement: Outperforms AlphaFold3 in prediction accuracy
- Pandemic-ready: Effective even with limited data on new viruses
- 33 targets identified: Found common factors across influenza strains
- Atomic precision: Generates detailed interaction models for drug design


